U.S. Reps. Peter Welch (D-Vt.) and David McKinley (R-W.Va.) have reintroduced bipartisan legislation in the U.S. House to give 340B rural and free-standing cancer hospitals access to 340B pricing on orphan drugs—which according to a 2019 study are 25
…Category: Federal
A second Republican 340B-related amendment, regarding what covered entities could charge patients for insulin, was offered and then tabled in the U.S. Senate late last week to a budget bill linked to Democrats’ forthcoming COVID-19 relief package.
Sen. Kevin Cramer’s
…Hikma Pharmaceuticals USA, the U.S. division of U.K.-based generic drug manufacturer Hikma Pharmaceuticals plc, is crediting 340B covered entities for overcharges on a long list of NDCs from the fourth quarter of 2017 through the fourth quarter of 2020.
The
…A Republican amendment offered, then tabled, in the U.S. Senate Feb. 4-5 to a budget bill linked to Democrats’ forthcoming COVID-19 relief package could have led to Senate action related to “improving transparency” in the 340B program “through data reporting.”
…The bipartisan group of six U.S. House members that got 217 total representatives last fall—173 Democrats and 44 Republicans—to sign a letter opposing efforts to replace up-front 340B drug discounts with back-end rebates began circulating a new 340B sign-on letter
…U.S. Senate Democrats late Tuesday afternoon announced their committee assignments for the 117th Congress. Senate Republicans had not yet announced their committee members as of this morning. However, Sen. Richard Burr (N.C.) will be the ranking Republican on the Health,
…The generic drug manufacturer recently required to repay covered entities for charging above the ceiling price following an audit has posted a notice on the U.S. Health Services and Resources Administration (HRSA) website about recalculation of ceiling prices and “potential
…The Biden administration and Pharmaceutical Care Management Association (PCMA), the trade association for pharmaceutical benefit managers (PBMs), have agreed to postpone, from January 1, 2022, to January 1, 2023, the effective date of the Trump administration rule eliminating safe harbor
…Drug manufacturer Sanofi has exempted 11 of the 16 types of health care providers eligible to participate in the 340B program from its conditions on 340B pricing for its products dispensed by contract pharmacies. However, consolidated health center programs (CH),
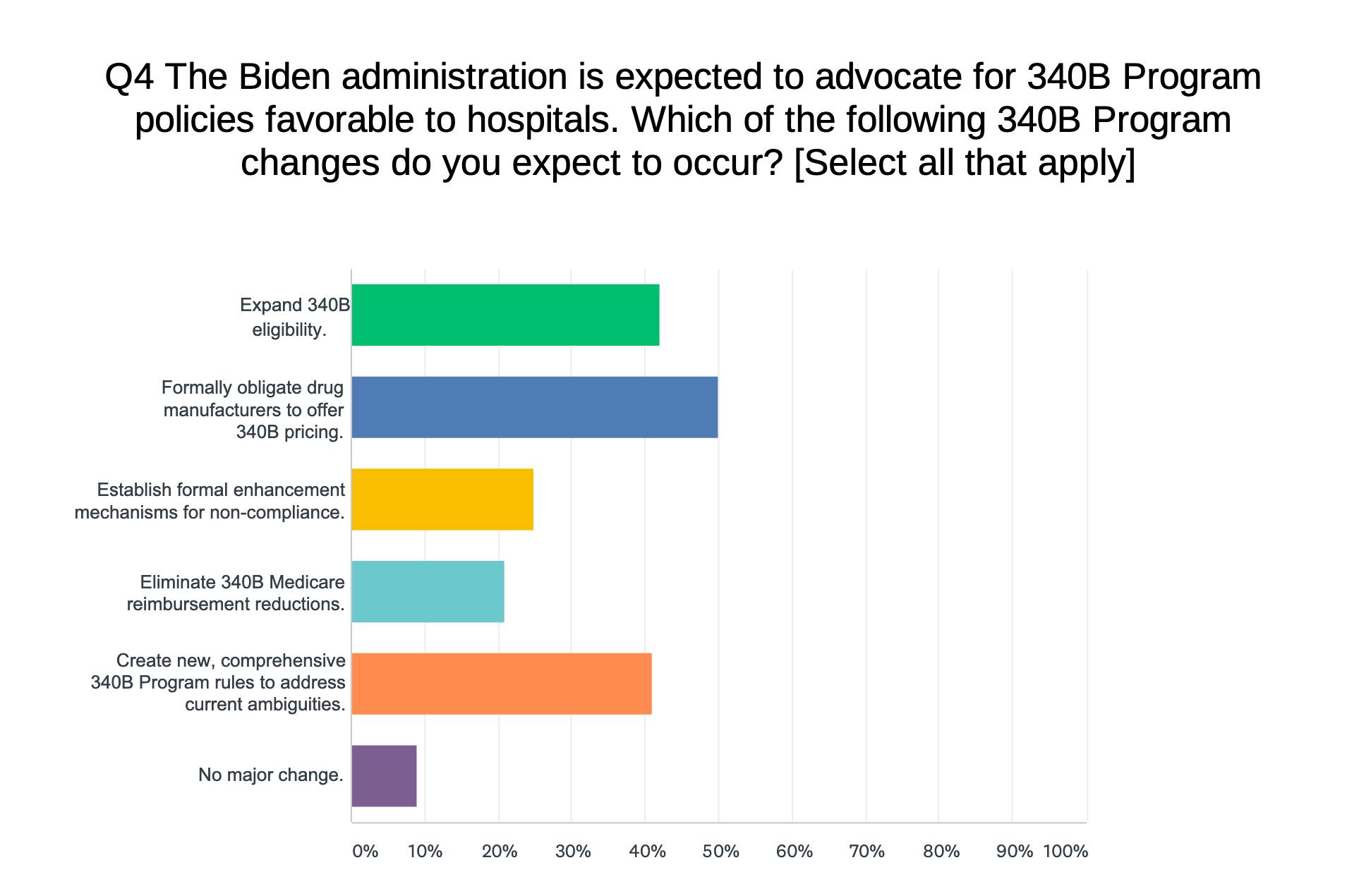
…Half (50 percent) of a sample of health care industry C-suite executives surveyed last month expect the Biden administration to “formally obligate drug manufacturers to offer 340B pricing,” consulting firm Advis reports in its first health care industry leadership survey
…