Many 340B hospitals “may not offer low-income patients financial assistance to access medicines and may use extraordinary collection actions, including but not limited to, liens, foreclosures, and civil actions when patients fail to pay bills,” a pharmaceutical-industry funded study released
…Category: Research/Reports
One-third (33%) of community health center (CHC) senior executives whose organizations do not currently have an on-site pharmacy plan to open one in the next three years, a new survey shows.
340B pharmacy management company Maxor 340B released the survey
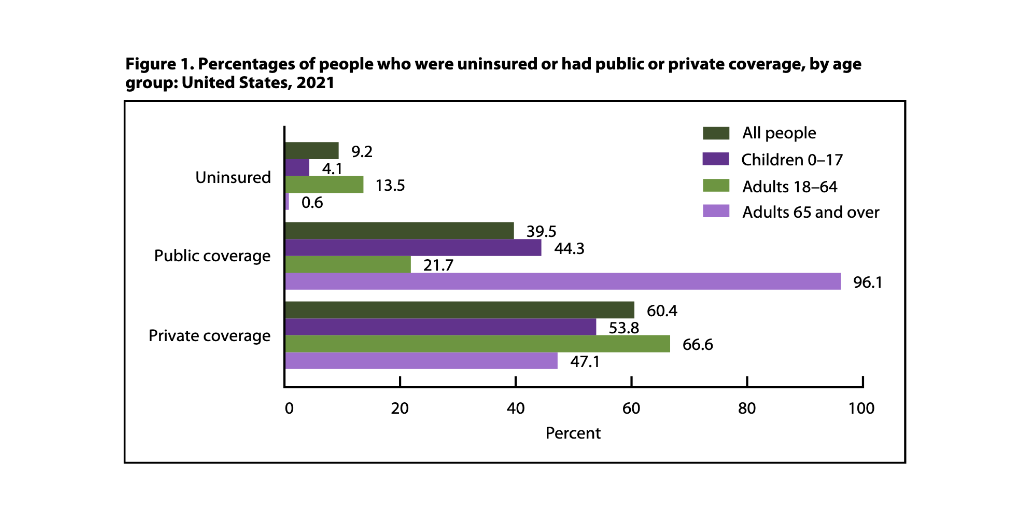
…The U.S. uninsured rate fell slightly in 2021 to 9.2% (about 30 million people), down from 9.7% (about 31.6 million people) in 2020, the National Center for Health Statistics (NCHS) reports.
NCHS released this and related health insurance coverage findings earlier
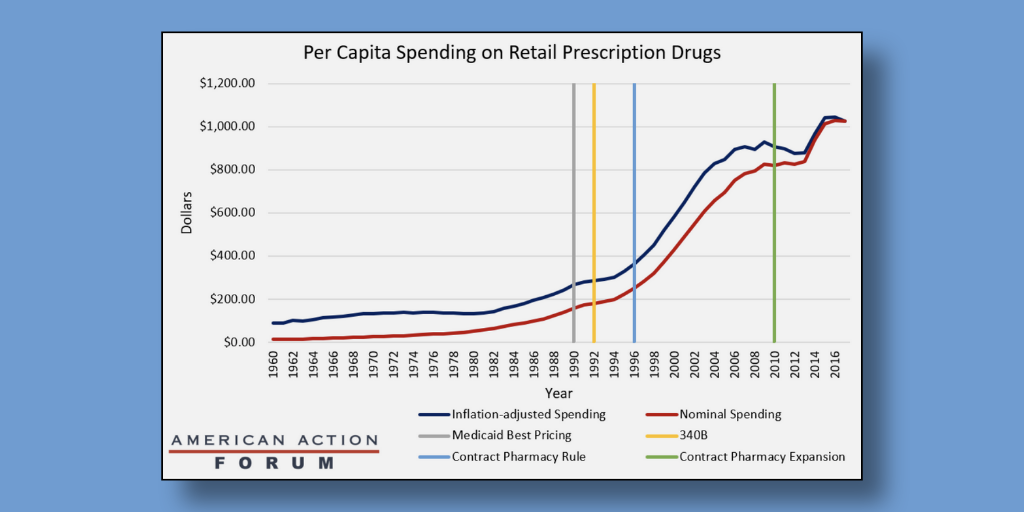
…An American Action Forum (AAF) data analysis that connects increases in consumer drug spending with the introduction of 340B drug discount and Medicaid drug rebate program policies “is spurious at best,” says an expert on prescription drug costs whose research
…The financial impact to 340B hospitals due to drug manufacturers’ restrictions on contract pharmacy more than doubled between December 2021 and March 2022, a new survey shows.
Hospital trade group 340B Health said the median annualized loss of contract pharmacy
…340B disproportionate share (DSH) hospitals’ contract pharmacies and child sites often are in richer and less diverse places where more residents have health insurance compared with the hospitals’ own locations, according to a new study.
Healthcare consulting firm Avalere Health
…Many National Cancer Institute (NCI) designated cancer centers—most of which are affiliated with 340B hospitals—mark up the price of infused cancer therapies for patients with private insurance “substantially,” according to new research in JAMA Internal Medicine. Hospital groups have pointed
…A nationally prominent drug policy expert and two colleagues at Harvard Medical School have written in JAMA how Congress could end the fight over 340B contract pharmacy and enhance 340B program integrity.
“First, Congress could explicitly allow the participation of
…Small rural hospitals have lost on average 39% of their 340B contract pharmacy savings due to drug manufacturers’ conditions on 340B pricing while larger, mostly urban hospitals have lost on average 23%, a hospital group’s member survey shows.
The finding
…Prescription drug sales at 340B-discounted prices grew 15.9% year-on-year from 2020 to 2021, drug industry consulting and contract research firm IQVIA reports, an annual growth rate below that for the two preceding years.
Last year, IQVIA reported that 340B sales
…