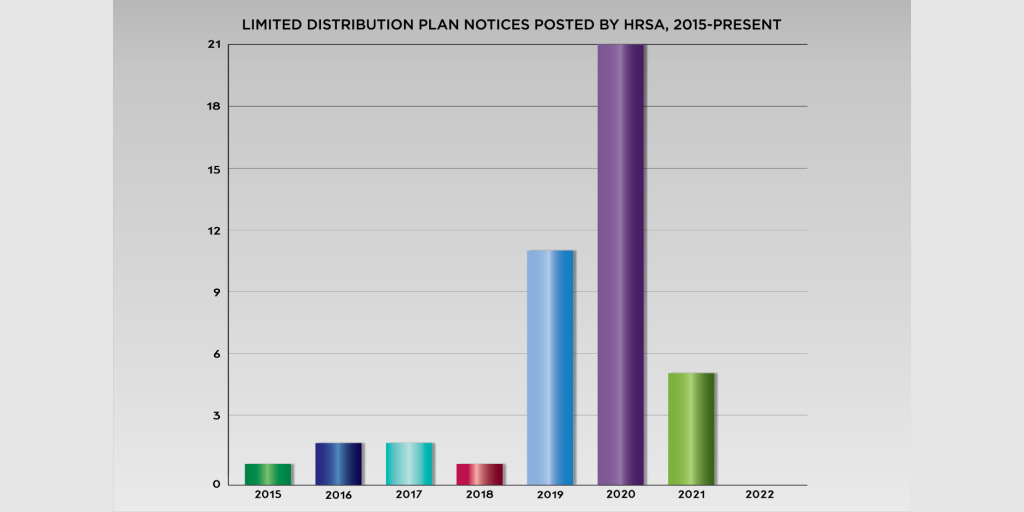
Generic drug manufacturer Unichem Pharmaceuticals USA must repay 340B covered entities for overcharges found during a 340B program compliance audit, the U.S. Health Resources and Services Administration (HRSA) said yesterday.
HRSA has been auditing five drug manufacturers per year for
…