The National Association of Community Health Centers urged Congress and the Biden administration this afternoon to force drug companies to ship 340B drugs to contract pharmacies unconditionally and stop pharmacy benefit managers from siphoning off health centers’ 340B savings.
NACHC called for new 340B-related laws protecting health centers and stronger enforcement of existing laws during a news conference to release findings from a survey of its members this spring. The survey focuses mainly on how manufacturers’ 340B contract pharmacy restrictions affect health centers and their patients.
This is not the first time that NACHC has called for federal legislation addressing manufacturers and PBMs’ 340B policies. The news conference, survey, and call for action, however, is a strong sign that the influential group thinks the situation is getting worse, and the government needs to step it up.
“Health centers are caught in the middle of corporate greed between pharmaceutical manufacturers and PBMs, both of whom want a larger piece of health centers’ 340B savings,” the survey report said. “Given the uncertain future of the 340B program threatens health centers’ financial viability, Congress and the administration need to implement solutions to address current issues and bolster protections to create a sustainable 340B program.”
NACHC urged Congress and Biden to:
- Pass legislation requiring manufacturers to ship 340B priced drugs to contract pharmacies without conditions or requirements. “It is critical for Congress to amend the 340B statute to expressly include contract pharmacies in the program,” NACHC said. Many 340B provider organizations have been reluctant to open the 340B law over concerns that they would have to provide concessions to the drug industry. However, with financial losses growing from the contract pharmacy restrictions, some provider groups are now considering the option of amending the 340B law.
- Pass the 340B PROTECT Act, which NACHC said “will prohibit health insurers and PBMs from redirecting health center 340B savings to their pocket” and address “manufacturers’ need for more transparency by utilizing a third-party neutral clearinghouse to review claims data to prevent Medicaid duplicate discounts.” The bipartisan House bill has 94 co-sponsors.
- Continue the 340B administrative dispute resolution process to address disputes between covered entities and drug manufacturers.
- Explore all possible enforcement actions against manufacturers. NACHC specifically cited imposition of 340B program civil monetary penalties and alluded to potential exclusion of manufacturers’ drugs from Medicaid reimbursement. The U.S. Health and Human Services’ Office of Inspector General is considering imposing CMPs on seven of the 17 the companies that HHS says are breaking the 340B law.
NACHC noted that 22 states so far have passed laws “prohibiting PBMs from discriminating against 340B pharmacies. More states should follow their example “to protect health centers while awaiting consideration of a federal solution,” it said.
“Unilateral actions by manufacturers and drug middlemen have removed needed resources from communities that need them the most,” said Mike Holmes, NACHC Board Chair and CEO of Scenic Rivers Health Services in rural Minnesota, in a news release about the survey.
“We need Congress to make it clear that patients come before profits,” Holmes said.
Survey Findings
NACHC surveyed 302 federally qualified health centers and look-alikes between April 11 to May 13, 2022, about how they use 340B contract pharmacies and 340B drug discounts.
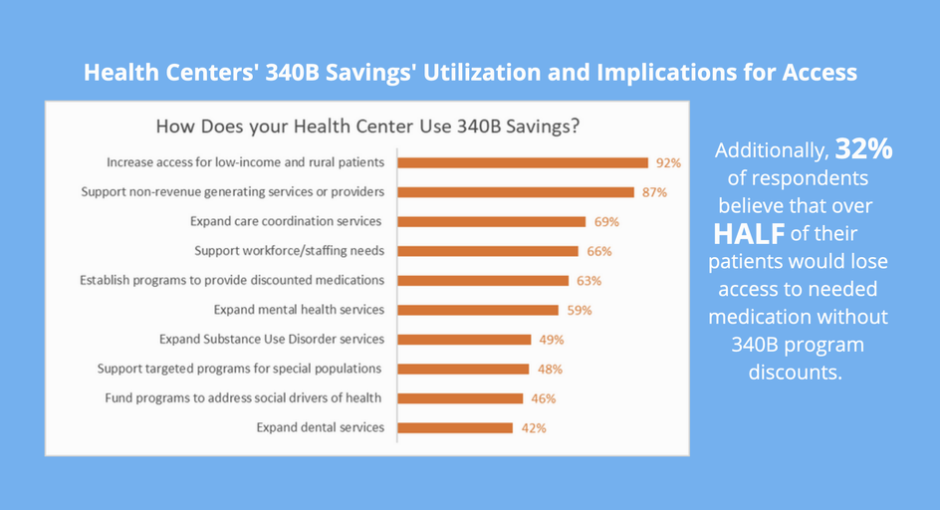
Thirty-two percent of the centers said more than half of their patients would go without needed medication if the center lost access to 340B drug discounts. Eighty-eight percent said at least 10% of their patients would lose medication access.
Eighty-six percent of the health centers surveyed said they used at least one contract pharmacy. Forty-one percent used less than 10, while 14% used more than 50.
Fifty-six percent of the health centers said they had an in-house pharmacy. (It was rare for centers to have one when the 340B program was created 30 years ago. Manufacturer restrictions on 340B contract pharmacy are prompting a growing number of health centers to consider opening one.)
The survey found that diabetes (96% of centers), heart disease (88%), and behavioral and mental health (56%) are the top three disease states treated with 340B-purchased drugs at health centers, followed by HIV/AIDS (18%), respiratory and reproductive health (12% each), and substance abuse (11%).
Ninety-two percent of health centers said they use 340B savings to maintain or expand services for low-income and/or rural patients in underserved communities. Eighty-seven percent said 340B savings subsidize services they otherwise could not afford to offer.
Eighty-nine percent said their participation in 340B improves medication adherence, 86% said it improves patient clinical outcomes and access to care, and 66% said it improved patient satisfaction.
NACHC provided 340B Report with embargoed pre-publication drafts of the survey report and news release. We will report tomorrow about NACHC’s news conference.