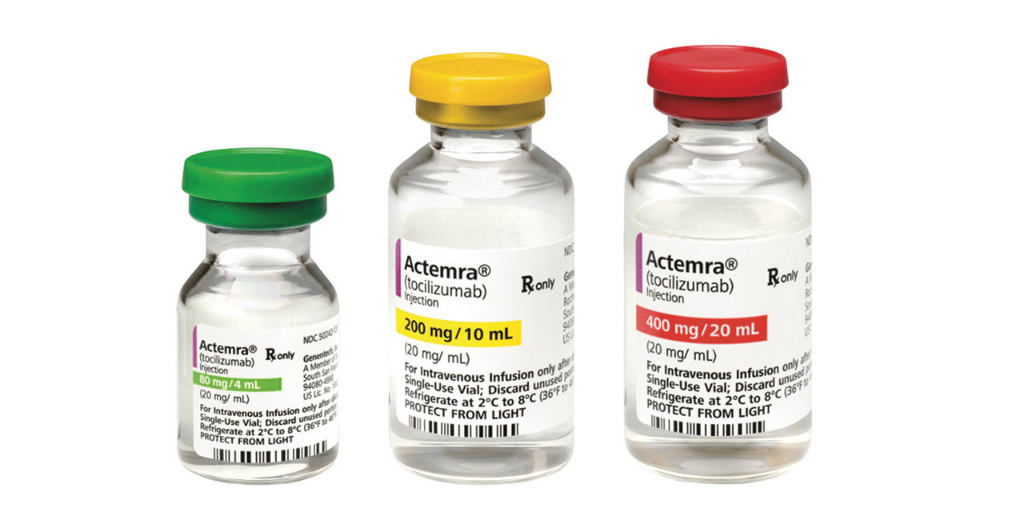
Biopharmaceutical manufacturer Genentech notified 340B entities this week that it is running out of Actemra IV (tocilizumab)—a key tool in treating hospitalized COVID-19 patients. It told entities they will be subject to the same allocation procedures as other buyers.
The
…